The Centers for Disease Control and Prevention (CDC) has reported 29 cases of severe allergic reactions to the Pfizer COVID-19 vaccine out of the nearly 5 million people who have already been vaccinated.
The report is the first that the CDC has released on allergic reactions following COVID-19 vaccinations. The findings have signaled to health officials that the Moderna and Pfizer vaccines that have been distributed don’t point to concerning trends, according to Business Insider.
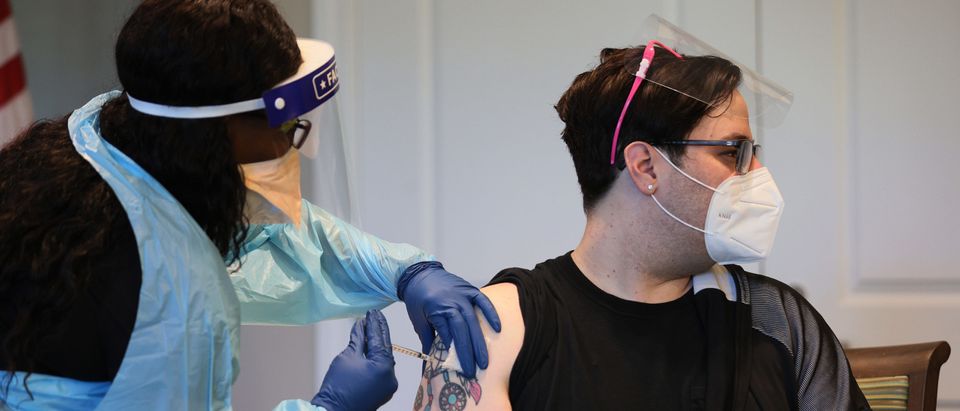
POMPANO BEACH, FLORIDA – JANUARY 06: A healthcare worker with American Medical Response, Inc working with the Florida Department of Health in Broward administers a Pfizer-BioNtech COVID-19 vaccine at the John Knox Village Continuing Care Retirement Community on January 6, 2021 in Pompano Beach, Florida. (Photo by Joe Raedle/Getty Images)
“Our vaccine safety systems haven’t picked up any worrisome signals,” Nancy Messonnier, a top CDC official told reporters Wednesday, accoridng to Business Insider. “This means that right now, the known and potential benefits of the current COVID-19 vaccines outweigh the known and potential risks of getting COVID-19.” (RELATED: EU Recommends Moderna Coronavirus Vaccine For Use)
The 29 people suffered anaphylaxis, a severe, potentially life-threatening allergic reaction that can be treated with epinephrine to mitigate the body’s allergic response.
At this rate, 5.5 people per every one million can be expected to develop an allergic reaction, which is higher than that seen in people who got the flu shot, according to the Associated Press.
Among the first 1.9 million people to have been vaccinated as of Dec. 23, 21 suffered allergic reactions, leading to 19 people receiving epinephrine while four were hospitalized. No one died, according to the CDC report. Since this initial report, 8 additional cases have been detected, totaling 29 nationwide.
Four of the 21 people who had anaphylaxis in December had a past history of allergies, while a majority of them had never experienced anaphylaxis before. Most reactions happened within 15 minutes of the vaccination.

LOUISVILLE, KY – JANUARY 04: A health worker administers a vaccine to a patient in their vehicle during the first day of mass Moderna COVID-19 vaccinations in Broadbent Arena at the Kentucky State Fair and Exposition Center on January 4, 2021 in Louisville, Kentucky. (Photo by Jon Cherry/Getty Images)
The CDC is keeping track of potential allergic reactions through a texting system that asks patients about systems after receiving the first of two shots. There is also an existing Vaccine Adverse Events Reporting System where people who have had reactions to the vaccine can submit an online form to the CDC and Food and Drug Administration (FDA) about their experience.