Americans who are not classified as priority recipients of the coronavirus vaccine by their state government will have an easy time skipping the line and receiving it early, according to health experts.
Groups classified as highest priority — such as those living in nursing homes and frontline health care workers — will be easy to inoculate with the coronavirus vaccine without any line-skipping, according Axios. However, local officials tasked with distributing the vaccine will have an increasingly more difficult time enforcing who can and cannot receive it as the distribution process progresses.
“Eventually you’ll get to the point where there’s a lot of providers and distribution points involved in this plan, and it’s going to be harder and harder to ensure you adhere strictly to these priority groups,” Josh Michaud, associate director of global health policy at the Kaiser Family Foundation (KFF), told Axios. “I’m sure there will be a point where we see line jumping.”
The Centers for Disease Control and Prevention formulated guidelines for vaccine distribution, which it recommended to states, according to a KFF report. State officials ultimately have the final say on who gets priority access to the vaccine. (RELATED: Should Minorities Get Priority For A Coronavirus Vaccine? Some Think So)
“As far as enforcement, states will try to direct vaccines as best as they can to reach the populations they want to, but once they’re at those distribution points, it’s hard to control this process,” he added,” Michaud said, according to Axios.
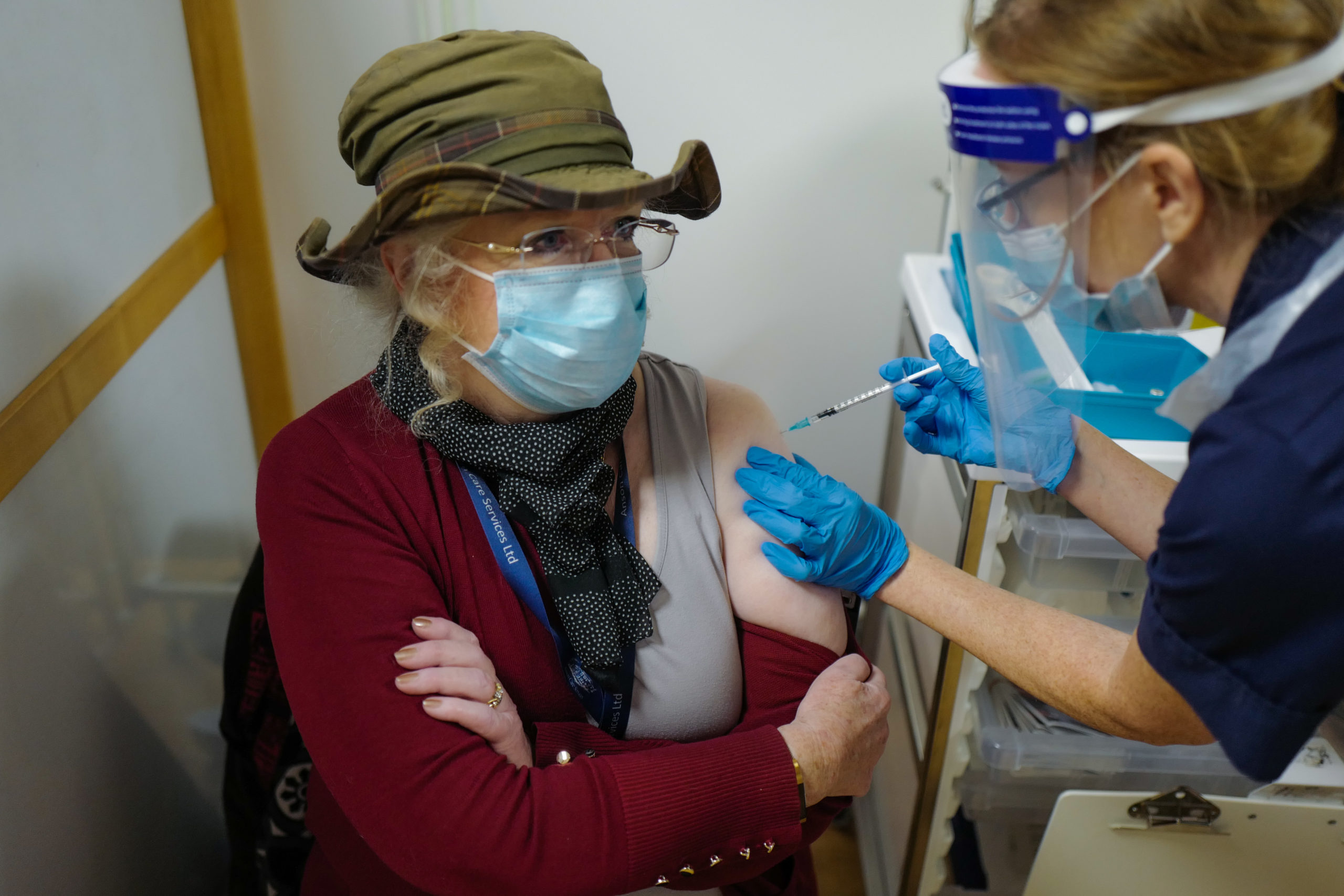
British citizen Mary Anson is among the first patients to receive the coronavirus vaccine on Dec. 9 in Truro, United Kingdom. (Hugh Hastings/Getty Images)
Pharmaceutical companies Pfizer and Moderna both recently requested emergency use authorization from the federal government for their coronavirus vaccine candidates after reporting positive large-scale human trial results.
A Food and Drug Administration (FDA) advisory committee recommended Pfizer’s vaccine for approval Thursday, The New York Times reported. The committee will reconvene on Dec. 17 to discuss Moderna’s vaccine.
The first inoculations could begin Monday, according to Health and Human Services Sec. Alex Azar.
“I doubt they’re going to require a lot of documentation,” Eric Toner, a senior scholar with the Johns Hopkins Center for Health Security, told Axios. “If you say you have diabetes, they’re not going to want to see your blood sugar.”
“I think we do have to depend to a significant extent on people’s honesty, and to some extent, it’s an honor system. You can check age…but the rest of it you really can’t,” Toner said. (RELATED: Coronavirus Vaccine Will Not Be Mandatory, Joe Biden Says)
But, there’s a silver lining if people do cut the line, according to Toner and Bryan Mroz, Acting Assistant Secretary of the Maryland Department of Health said, Axios reported. With a significant portion of the population expressing doubts about the efficacy of the vaccine, Toner and Mroz said cutting the line will simply ensure more people are getting the vaccine sooner.
“That’s a problem I kind of want to have — people lined up to get [vaccinated],” Mroz told Axios.
Average coronavirus cases and deaths per million have been increasing steadily in the U.S. since mid-October, according to The COVID Tracking Project. On Thursday, the U.S. reported 3,067 new coronavirus-related deaths and 215,669 new cases while 107,258 Americans remained hospitalized from the virus.
All content created by the Daily Caller News Foundation, an independent and nonpartisan newswire service, is available without charge to any legitimate news publisher that can provide a large audience. All republished articles must include our logo, our reporter’s byline and their DCNF affiliation. For any questions about our guidelines or partnering with us, please contact licensing@dailycallernewsfoundation.org.












